
Multiply Labs builds robotic systems that automate your existing biomanufacturing instruments with no process transfer, no lock-in. Our technology brings closed-system GMP automation to your full, end-to-end process. Your scientists supervise, discover, and innovate while the robots execute inside the cleanroom.
Each cluster packs four robotic arms and twelve modular instrument towers into under 600 square feet, compatible with the GMP instruments your team already uses. No process modifications. No lock-in.
The robots execute your manufacturing process inside a closed, GMP-compliant environment, supporting sterile conditions, significantly reducing contamination risk, and delivering reproducible results run after run.
The Best Part:
You can always customize the manufacturing system to fit your process.

The cluster integrates robotic execution, instrument orchestration, and process intelligence into a single closed system.

The Process Creator App (PCA) is a drag-and-drop, no-code interface that lets your scientists build, modify, and own your manufacturing process.

Robots are adapted to work with your existing equipment, automating the touchpoints so your team can focus on process development and oversight.

Four Universal Robots cobots execute liquid handling, dispensing, and transfer steps inside a closed, sterile environment.

Purpose-built software collects process data (every step, sensor reading, and decision point) and delivers complete electronic records directly into your quality system.
You've spent years developing your manufacturing process on the GMP instruments your team trusts. We don't ask you to change them. Our robotic infrastructure is adapted to work around your existing equipment, automating the touchpoints, reducing manual handling, and delivering consistent outcomes at scale.

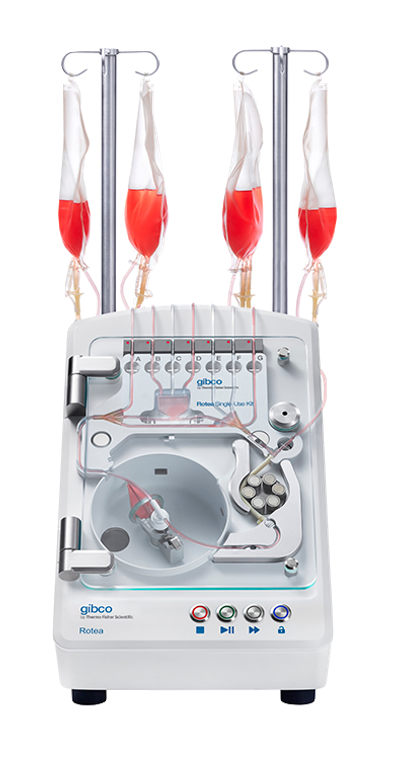

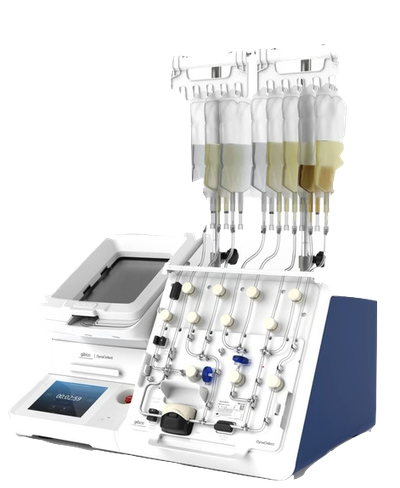












Advanced biologics manufacturing comprises dozens of discrete unit operations, each one requiring a highly controlled, closed sterile environment and strong repeatability across every transfer, every dispense, every interaction with the material. Multiply Labs automates the entire sequence.
50+ unit operations. All robotic. All inside a closed, GMP-compliant system.


Every consumable is purpose-engineered for robotic handling: precise locating, secure retention, and closed transfer. Load them in, and the robots do the rest.








Multiply Labs' software translates your manufacturing protocol into robotic execution, collecting every sensor reading and every interaction in real time, delivering complete electronic records directly to you and your quality system.
Tell us about your manufacturing program and we'll schedule a personalized walkthrough tailored to your therapy type and scale goals.
Request an In-Person Visit